METALLIC ALLOYS
Home » Disturbance fields » METALLIC ALLOYS
It has been scientifically proven that various metals such as amalgam, gold, platinum, copper, cobalt, aluminium, iron and chromium can have a toxic effect on cells, an effect on immunology, a carcinogenic effect and an effect on metabolism.
Neither amalgam nor any of the metals used in dental alloys is a natural component of the body.
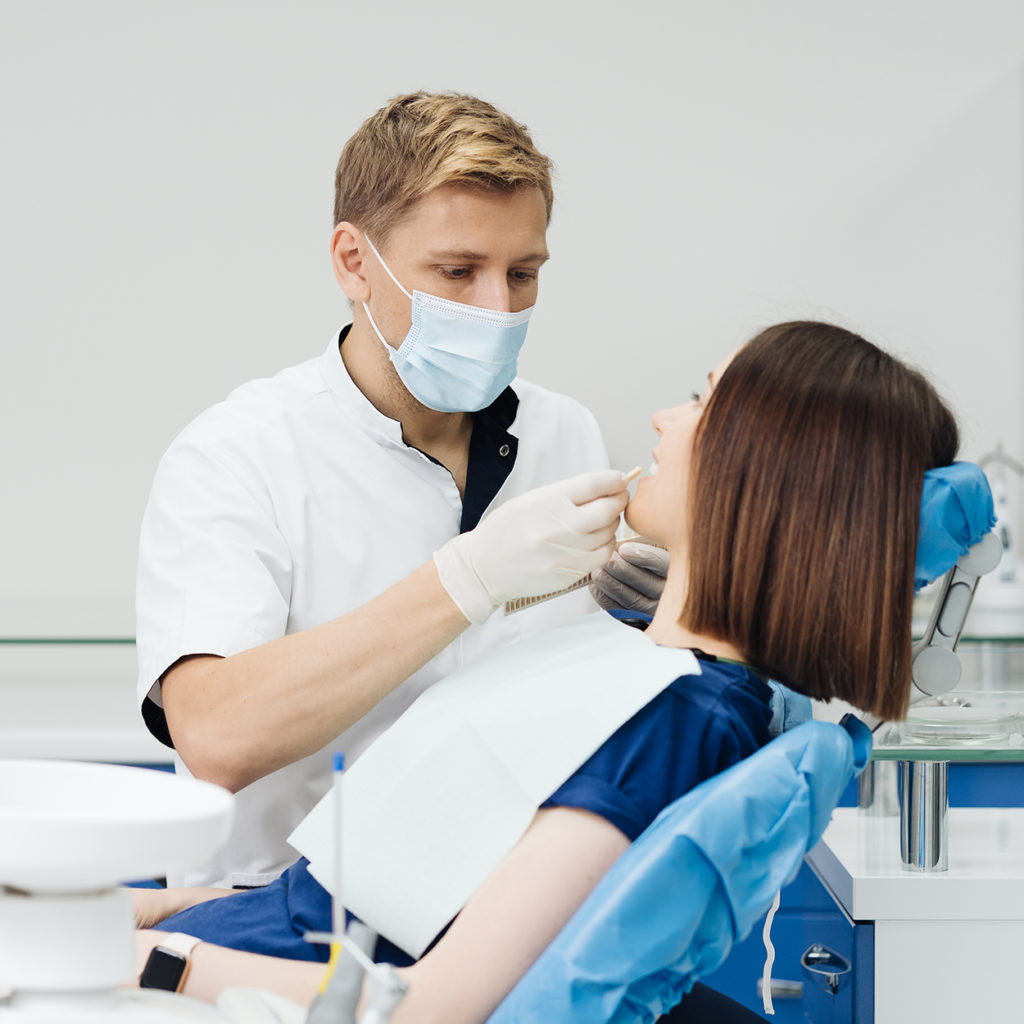
The toxicity of amalgam
Amalgam is an alloy of silver and mercury which is classified as a heavy metal and is highly toxic. Amalgam is increasingly banned or contraindicated throughout the world because of its toxicity. This is described in the scientific literature in three ways:
- Toxicity in relation to mercury in the form of vapours.
- Toxicity in relation to mercury in the form of methylmercury salts.
- Toxicity in relation to electromagnetic currents and fields generated by its metallic composition. The presence of several metals in the mouth causes a battery-like current known as electrogalvanism.
This electric current can lead to parafunctions such as bruxism (teeth grinding) or tinnitus. A few days after they have been inserted in the mouth, traces can be found in the rest of the body.
The harmful effects of metals
Basically, we can distinguish three harmful effects of metals:
Here, it is amalgam and its toxicity that plays a particular and decisive role, with its components mercury, copper, tin and silver.
No metal used in dentistry has a role to play in the human body. In practice, each metal stimulates the immune system like a foreign body and can even cause an allergic problem. This process is totally independent of the quantity of metal, the number of metal crowns, inlays or titanium implants. It can play a role in the development of autoimmune diseases such as multiple sclerosis and Hashimoto's.
With today's cellular phones, Wi-Fi and other wireless networks, we are exposed to a large number of electromagnetic frequencies and waves. Metals placed in the mouth can act like small antennae and damage the nervous system. The waves can cause a rise in temperature in the surrounding tissues. The effects on the body are uncontrollable.
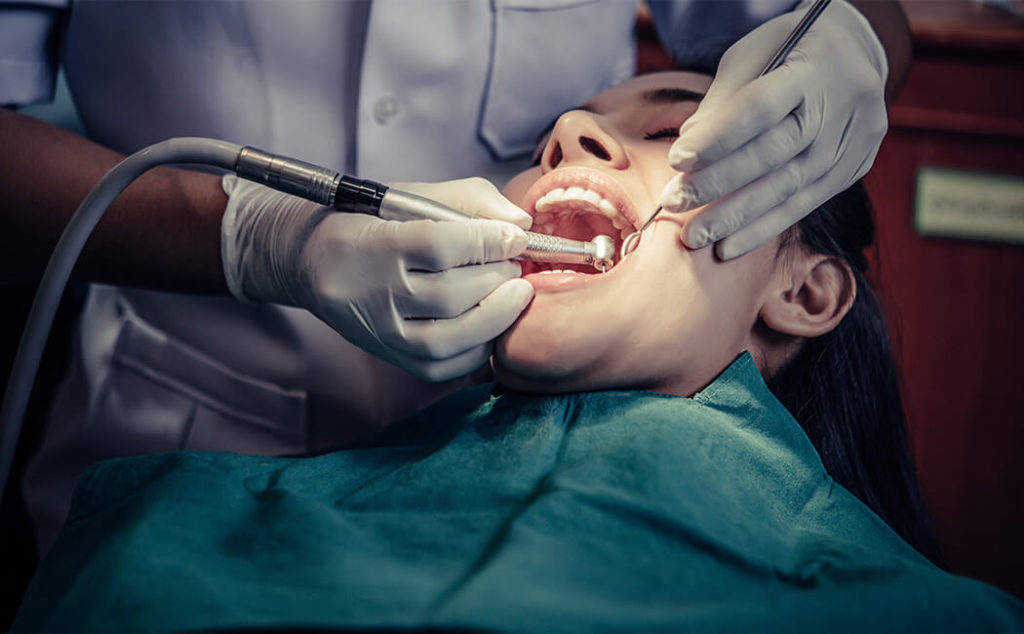
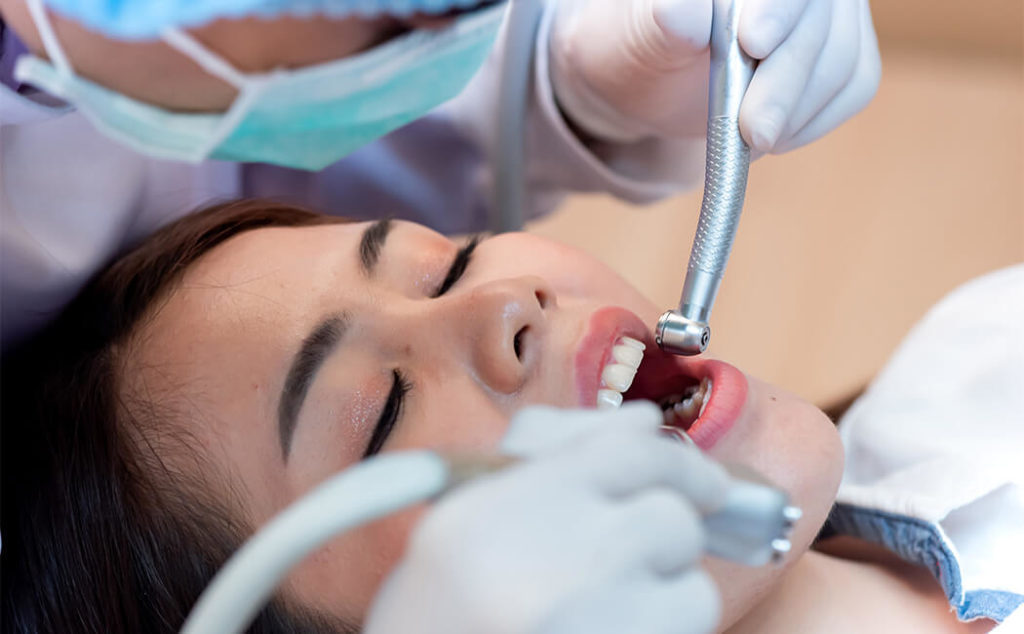
Protection of the body during the removal of metals
To prepare the body in the best way possible for the removal of metals, you will begin our suggested regime 14 days before the appointment, or continue with the regime suggested by the referring doctor or naturopath.
